(Photo
source: WLOX, Google Maps)
Posted: Aug 13, 2015
By Cassie Archebelle
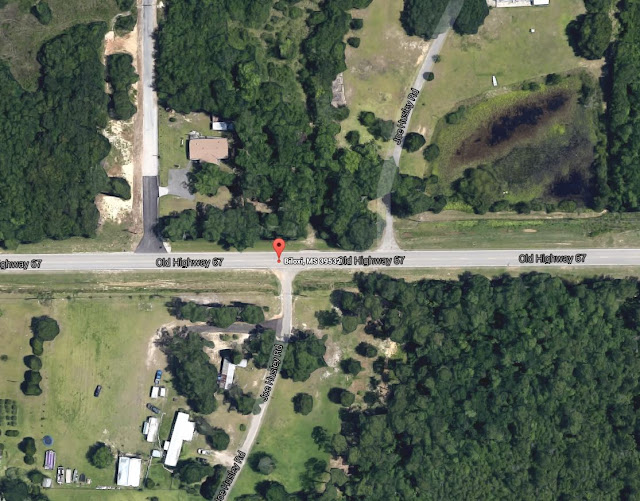
BILOXI (WOOL MARKET), MS (WLOX) -
Two hazmat certified firefighters made quick work of a
chlorine tank leak at a Harrison County water well Thursday afternoon. The leak was reported around 1:30 p.m.
Biloxi fire officials said the tank was in the Wool Market
community near the intersection of Old Highway 67 and Joe Husley Road.
After securing the leak, the firefighters were checked out
and given the all clear by AMR personnel.
Since then, Hazmat teams have repaired the leak.
No injuries were reported.
//-------------------------///
Orientation and General Overview:
The City of Biloxi has twenty three (23) water well sites that are utilized to
produce drinking water for the citizens and businesses of the City. Fifteen
(15) of these water wells have two (2) cylinders each of liquid chlorine, each
of which contains 2,000 pounds, for a total of 4,000 pounds of liquid chlorine
at each well site. This RMP is for one of those fifteen (15) water well sites.
The City of Biloxi operates and maintains the City's water production and
distribution system, and its wastewater collection system. The City owns the
utility facilities and is therefore responsible for the submission of this RMP.
Nature of Facility Risk Imposed and Risk Minimization:
The primary facility risk to the public is that liquid chlorine could be
released into the atmosphere as a toxic gas (a vapor). A total rupture of the
chlorine cylinders or a total failure of the process equipment is highly
unlikely; therefore a sudden and total release of all of the chlorine is also
highly unlikely. The more likely scenario is the slow release of chlorine due
to a leak of some type. Although great measures have been taken to prevent such
leak event, such leak would result in the dispersal of chlorine gas to about .1
mile from the facility, according to the RMP*Comp Model, Version 1.06. To
minimize this risk, numerous steps and measures have been taken and documented
throughout this Executive Summary and the RMP. A specific example not noted in
the RMP is the change-out of the chlorination system from pressure regulators
to vacuum regulators, which is a far safer and more reliable system.
RMP*Submit - Data Entry Explanations:
Section 1.11 asks the question "Number of full time employees on
site". The described facility is inspected every single day of the year by
a city employee that holds a current and valid Waterworks Operator
Certification as promulgated by the State of Mississippi Department of Health.
Each daily inspection generally takes
20 to 30 minutes. The data field will not accept a "fraction of an
employee" expressed in a decimal value. The EPA hotline attendant for the
RMP program suggested that a "1" be entered in this data field and
then explained with an entry in the Executive Summary, which this paragraph
accomplishes.
Below are summaries of selected elements of the RMP, as specifically requested
in the RMP User's Manuals, Pages 2.1 through 2.2:
Accidental Release and Prevention Emergency Response Policies:
At this facility, chlorine is handled, which is considered by the EPA to be a
hazardous material. The same qualities that make chlorine valuable as a commodity
also make it necessary to observe certain safety precautions in handling
chlorine to prevent unnecessary human exposure, to reduce the threat to the
personal health of persons working with and around the chlorine, and to reduce
the threat to nearby members of the community.
It is the policy of the City to adhere to all Federal and state rules and
regulations. Safety depends upon the manner in which the chlorine is handled,
the safety devices inherent in the design of the facility, the safe handling procedures
that are used, and the training of personnel.
The accidental release prevention and emergency response policies were modeled
from various professional sources, such as the Chlorine Institute's Pamphlet
64, Emergency Response Plans for Chlorine Facilities, J. J. Keller and
Associates, Inc.'s Keller-Soft Safety Plans, and OSHA documentation.
Facility and the Regulated Substances Handled:
Two (2) cylinders of liquid chlorine, containing 2,000 pounds each, are located
at this facility, for a total of 4,000 pounds of liquid chlorine.
The primary purpose of this facility is to distribute chlorine into the public
water system in a safe and reliable manner that is in conformance with
regulations as established by the Mississippi Department of Health, the
Mississippi Department of Environmental Quality, and the U. S. Env
ironmental Protection Agency. Two (2) cylinders are maintained at the facility
to ensure a constant supply of chlorine when one of the tanks becomes empty.
The chlorine is provided by a licensed and certified vendor and is delivered in
vehicles that are in conformance with Department of Transportation regulations.
A chain link fence that allows visibility secures this facility. This fence is
chained and locked. There is also another chain link fence immediately
surrounding the chlorine cylinders. It is also chained and locked. The grounds
are mowed and maintained in such a manner that the facility is in full view and
easily accessible by authorized personnel. The site is restricted to authorized
facility employees, authorized management personnel and authorized contractors
only. There are signs clearly posted (including a telephone number) for
notification in the event of an emergency
There are established written procedures for changing chlorine cylinders, as
follows:
CHANGING A CHLORINE CYLINDER
STEP 1: Notify dispatch to advise your location and that you are changing a
chlorine cylinder, and the size of the cylinder.
STEP 2: There must be two certified operators present to change a chlorine
cylinder.
STEP 3: The operators must use Self-Contained Breathing Apparatus (SCBA) when
changing chlorine cylinders.
STEP 4: Turn the well on manual to ensure that it remains running. Wait until
the pump starts and the chlorine booster pump is running.
STEP 5: Turn the chlorine cylinder off.
STEP 6: Check the rotameter to ensure that it drops to zero and stays there. If
the rotameter does not drop to zero, perform maintenance as needed on the poly
tubing to ensure that the rotameter drops to zero.
STEP 7: With the well still running, slowly loosen the regulator. Do not take
it off immediately. If no chlorine is released, remove the regulator yolk.
STEP 8: Check the valve on the new replacement cylinder to assure that it is
off. Slowly loosen the valve cap on the replacement chlorine cylinder. Check
for leaks using an ammonia solution. If there is a leak, tighten the cap to
stop the leak and notify dispatch to notify the vendor to retrieve the cylinder
and leave a replacement cylinder. If there is no leak, then remove the cap.
Then clean the face of the valve outlet.
STEP 9: Remove the lead washer from the yolk. Clean the area and always use a
new washer!
STEP 10: Put the yolk over the cylinder and tighten it. Make sure that the
nipple fits properly in the socket. Do not over-tighten the nipple.
STEP 11: Open slowly the cylinder valve and check for a leak. If there is a
leak, close the valve and slightly tighten the yolk assembly. Then slowly open
the valve and check again for a leak. Repeat this procedure until there is no
leak. If there still is a leak and this procedure does not stop it, then
tighten the cap to stop the leak and notify dispatch to notify the vendor to
retrieve the cylinder and leave a replacement.
STEP 12: Turn the well back to the automatic mode.
STEP 13: Record in the daily logbook the date, time, personnel present and the
maintenance performed.
STEP 14: Call dispatch to notify them that you have completed changing the
cylinder.
Worst -Case Scenario and Alternate Release Scenario:
Worst-Case Scenario - Total failure of either or both cylinders is highly
unlikely. However, if such an event did occur, then 4,000 pounds of liquid
chlorine could be released into the atmosphere as a toxic gas (a vapor), which
is heavier than air. According to the RMP*Comp Model, Version 1.06, the
distance to the endpoint would be 1.9 miles. Based on guidelines as published
by The Chlorine Institute, Inc., this scenario assumes a release rate of 400
pounds per minute for release duration of 10 minutes, wind speed of 3.4 miles
per hour, air temperature of 77 degrees, air stability class F, no mitigation
measures and urban surroundings.
Alternate Case Scenario - A malfunction occurs during a cylinder change-out and
there is a slow release of liquid chlorine into the atmosphere as a toxic gas
(a vapor), which is heavier than air. Although also remote, this scenario was
selected because it is probably the most likely incident to occur that would
cause a release of chlorine gas. Based on guidelines as published by The
Chlorine Institute, Inc., this scenario assumes a release rate of 10 pounds per
minute for release duration of 400 minutes, wind speed of 6.7 miles per hour,
air temperature of 77 degrees, air stability class D, no mitigation measures
and urban surroundings. Under this scenario, according to the RMP*Comp Model,
Version 1.06, the distance to the endpoint would be .1 mile.
General Accidental Release Program and Chemical-Specific Prevention Steps:
This facility complies with the EPA's Accidental Release Prevention Rule and
with all applicable state codes and regulations, including the rules and
regulations as promulgated by the EPA, Mississippi Department of Health,
Mississippi Department of Environmental Quality, and OSHA. The facility also
follows guidelines as established by the American Water Works Association and
the Chlorine Institute.
Five Year Accident History:
There have been no accidents to report at this facility within the last five
years.
Emergency Response Program:
This facility's Emergency Response Program is based on the Chlorine Institute's
Pamphlet 64, Emergency Response Plans for Chlorine Facilities. The program
addresses the requirements of 40CFR 68 pertaining to notification, medical
treatment, response procedures, planning, use and maintenance of emergency
equipment, employee safety training, and emergency response evaluation. This
program has been discussed, reviewed and coordinated with the City of Biloxi
Fire Department. A representative of the City of Biloxi Fire Department visits
the facility on an annual basis, at a minimum, to inspect the facility for fire
prevention and overall safety. There is a formally designated and documented
Chlorine Emergency Response Team (CERT). The team is highly trained, performs
periodic practice drills, and is on notice 24 hours a day, 365 days a year. In
the event of an emergency, the City of Biloxi Fire Department in cooperation
with the City of Biloxi Police Department notifies the public.
Planned changes to improve safety.
Biloxi will continue to provide in house and out sourced training to all
employes who deal with chlorine.
 United States of America
United States of America